研究成果
○原著論文(2025〜 / 2024 / 2023 / 2022 / 2021 / 2020 / 2019 / 2018 / 2017 / 2016 / 2015 / 2014 / 2013 / 2012 / 2011 / 2010 / 2009 / 2008 / 2007 / 2006 / 2005 / 2004 / 2003 / 2002)
○総説・著書
2026
● Mandelic Acid-Mediated Enantiomeric Enrichment of a Resorcinarene-Based Dissymmetric Coordination Capsule
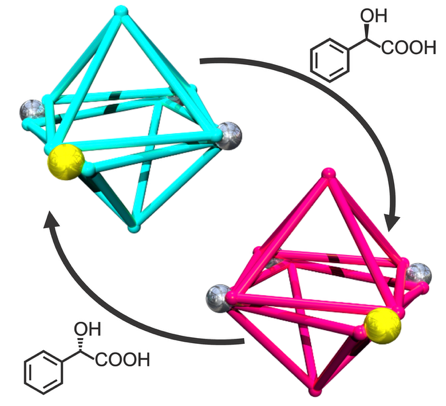
Resorcinarene-based coordination capsules provide rigid, well-defined cavities that can exhibit helicity under chiral induction, a feature that has previously been exploited using axially chiral biphenyl compounds and tartaric acid derivatives. However, these approaches generally require well-designed templates tailored to the capsule structures. In this study, it was discovered that commercially available mandelic acid functions as an effective and operationally simple additive for inducing chirality in an empty-cavity Cu(I) capsule without the requirement for templates. Chiral induction was achieved by introducing mandelic acid through either treatment of a preformed racemic capsule or during its assembly. Both approaches generated helically biased capsules, as confirmed by circular dichroism and nuclear magnetic resonance (NMR) spectroscopy, with optical purities approaching unity (up to ~97% ee) under optimized conditions. Mechanistic investigations using NMR spectroscopy, electrospray ionization mass spectrometry, and electron spin resonance spectroscopy indicated that the oxidation of Cu(I) to Cu(II) played a key role in the induction process, and that this was mediated by interactions with the carboxylate group of mandelic acid. This study therefore presents a straightforward and highly effective protocol for accessing an enantiomerically enriched resorcinarene-based coordination capsule, expanding its potential as a confined chiral reaction space and a functional chiral material.
Senri Miyagi, Yudai Ono, Manabu Abe, Tomoko Amimoto and Takeharu Haino, Chem. Asian. J., 2026, in Press
● Oligo(ethylene Oxide)–Polyoxometalate Macrocycles
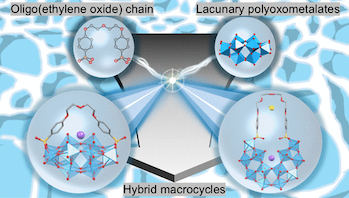
Host–guest chemistry has predominantly advanced within organic systems, whereas the integration of inorganic frameworks into such architectures remains limited. Here, we report two types of organic–inorganic hybrid macrocycles synthesized by cyclizing lacunary Wells–Dawson–type polyoxotungstates with oligo(ethylene oxide) chains. The resulting macrocycles exhibit distinct cavity structures depending on the chain length and show significantly enhanced hydrolytic stability in aqueous solution. The more stable host enables size-selective ion-exchange of the encapsulated K+ ion with alkylammonium cations, whose exchanged forms were crystallographically characterized. Thermodynamic analyses revealed enthalpy–entropy compensation across the guest species, indicating a common binding mechanism governed by desolvation and host conformational changes. This strategy demonstrates a versatile approach for constructing robust hybrid hosts that combine the flexibility of organic macrocycles with the rigidity of inorganic clusters.
Hiroto Kaitori, Jun Manabe, Fuga Onishi, Naoyuki Hisano, Takeharu Haino, Ryo Tsunashima, Takayoshi Nakamura, Sadafumi Nishihara, Chem. Mater., 2026, in Press
DOI:10.1021/acs.chemmater.6c00225
● Molecular recognition by a tris(phenylisoxazolyl)benzene derivative under flow-through conditions
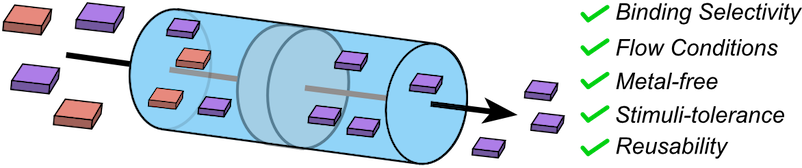
The application of host molecules as separation media has attracted considerable attention owing to their intrinsic molecular recognition capabilities. Herein, we report the host–guest complexation behavior of a tris(phenylisoxazolyl)benzene derivative under flow-through conditions. After thermal activation, the tris(phenylisoxazolyl)benzene derivative preferentially retains cis-decalin under flow-through conditions in the presence of octane, mesitylene, and trans-decalin to afford >95% removal of cis-decalin in the early-eluting fractions. Even after most of octane, mesitylene, or trans-decalin had eluted, cis-decalin remained retained in the column over the measured elution range. Calculations using the dimeric cavity of the tris(phenylisoxazolyl)benzene derivative provides a plausible structural rationale for the observed selectivity trends. Practically, this derivative maintains cis-decalin removal performance under acidic and basic conditions and after 365 nm irradiation. Moreover, it can be regenerated with no discernible performance loss over at least ten adsorption–regeneration cycles, highlighting its chemical/photochemical tolerance and reusability. This study underscores the potential advantages of the metal-free architecture of the tris(phenylisoxazolyl)benzene derivative, which maintains separation performance under acidic/basic conditions and UV irradiation. This may lead to the deployment of the tris(phenylisoxazolyl)benzene derivative and related non-macrocyclic hosts as scalable, selective media for flow-through separations of hydrocarbon mixtures.
M. Kawasaki, Y. Ono, T. Amimoto, T. Hirao, T. Haino, Adv. Sens. Res., 2026, in Press
● Noncovalent Synthesis of Amphiphilic Block Copolymers Through Host–Guest Interactions
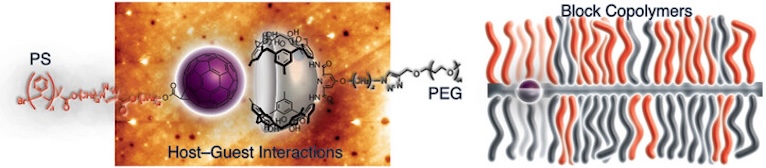
A supramolecular approach for constructing amphiphilic block copolymers has been developed based on host–guest interactions between calix[5]arene-functionalized polyethylene glycol (PEG) and [60]fullerene-functionalized polystyrene (PS). Supramolecular amphiphilic polymers were formed via spontaneous assembly in solution, as confirmed by UV/vis titration and DOSY NMR spectroscopy. Drop casting of a mixture of host- and guest-appended polymers onto substrates yielded smooth, continuous films. Atomic force microscopy (AFM) revealed, in addition to the smooth topographies, an alternating modulation in mechanical stiffness, which was ascribed to the interspersed PEG- and PS-rich regions. Differential scanning calorimetry (DSC) revealed transitions derived from the PEG and PS chains for a mixture of host- and guest-appended polymers, suggesting the coexistence of the PEG- and PS-rich regions. These results suggest that host–guest interactions successfully couple hydrophilic and hydrophobic polymer chains, suppressing macroscopic phase separation and yielding films with nanoscale mechanical heterogeneity. This strategy offers a modular and efficient platform for the fabrication of supramolecular amphiphilic block copolymers with tunable nanostructures.
Takehiro Hirao, Yuki Okishio, Ayako Ema, Masaya Yoshida, Shin-ichi Kihara, Takeharu Haino, Chem. Asian J., 2026, 21, e70691.
● Fluctuation-Driven Switching of Magnetic Exchange and Charge Transport in Ni(dmit)2 Salts with [24]crown-8-Based Supramolecular Cations
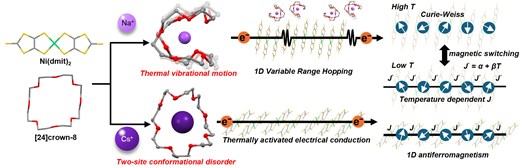
Countercation dynamics play a crucial yet largely unexplored role in governing the electronic properties of molecular conductors. Herein, we demonstrate that the fluctuations of flexible supramolecular cations in [Ni(dmit)2]-based molecular conductors (dmit2− = 2-thioxo-1,3-dithiol-4,5-dithiolate) can directly modulate their correlated electronic states. Two salts with nearly spherical supramolecular cations featuring Na+ and Cs+ ions encapsulated by unsubstituted [24]crown-8, Na([24]crown-8)[Ni(dmit)2]3 and Cs2([24]crown-8)2[Ni(dmit)2]7, are synthesized and systematically compared. Detailed crystallographic, magnetic, and transport studies reveal a striking contrast between the two systems. In Na([24]crown-8)[Ni(dmit)2]3, the structural distortion of the Na+-encapsulating [24]crown-8 enables thermally activated large-amplitude out-of-plane fluctuations, thus selectively enhancing the vibrational motion of terminal S atoms in [Ni(dmit)2] layers and leading to the temperature-dependent modulation of transfer integrals, anomalous magnetic behavior requiring a temperature-dependent exchange interaction, and one-dimensional variable-range hopping transport. In contrast, Cs2([24]crown-8)2[Ni(dmit)2]7 features a planar-confined Cs+-based supramolecular cation and suppressed fluctuation transfer to anion layers, thus exhibiting conventional Heisenberg-chain magnetism and Arrhenius-type semiconducting behavior. These results establish supramolecular-cation dynamics as a powerful design parameter for controlling electronic correlation and transport in molecular conductors.
Daisuke Ishikawa, Masato Haneda, Jun Manabe, Daisuke Shimoyama, Takeharu Haino, Kiyonori Takahashi, Takayoshi Nakamura, Sadafumi Nishihara, Bull. Chem. Soc. Jpn., 2026, uoag022.
● pH-Dependent Optical Properties of Nanographenes
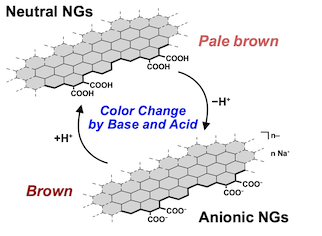
Nanographenes (NGs) produced by oxidative cleavage of carbons exhibit pH-dependent optical properties. We performed UV- vis and PL measurements on size-separated NGs across various pH levels and observed their halochromic behaviors in both the visible and near-infrared (NIR) regions. The absorption spectra exhibited a two-step change. The initial change is attributed to the deprotonation and protonation of the carboxy groups, and the latter is likely due to the aggregation and disaggregation of NGs. Notably, no significant shifts in the absorption and PL bands were observed, indicating that the deprotonation of the carboxy groups has a minimal impact on the electronic structures of NGs in both their ground and excited states. This implied that the effect of the deprotonation was limited to the edge moieties. Computational calculations suggested edge-to-edge and edge-to-surface π−π* transitions in the visible and NIR regions for a deprotonated model NG. Thus, a plausible explanation for the halochromic behavior of NGs involves the disappearance and appearance of these transitions upon the protonation and deprotonation of the carboxy groups. These changes affect the absorption in the visible and NIR regions, resulting in the halochromic behavior of NGs.
Mayu Nakamura, Kaito Fukushima, Aoto Ikeda, Kaori Miyazaki, Shun Yamaguchi, Yudai Ono, Takeharu Haino, Naoto Nishiyama, and Ryo Sekiya, ChemPhysChem, 2026, 27, e202500746.
● Structural phases in Ca2+-triggered alginate assembly and gelation: circular dichroism-guided multimodal analysis
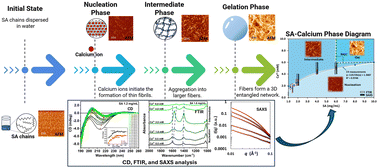
This study investigates the assembly and gelation behaviour of sodium alginate (SA) induced by calcium ions (Ca2+), using circular dichroism (CD), FTIR, SAXS, and AFM analyses. CD spectroscopy proved to be a highly sensitive and straightforward technique for detecting conformational changes and enabled construction of SA–Ca2+ phase diagram, revealing three distinct phases: nucleation, intermediate, and gelation. A red shift in the CD spectra in the 200–220 nm range indicated structural changes and the formation of “egg-box” structures. The CD spectra precisely pinpointed the nucleation-to-intermediate transition points across SA concentrations (1.0–6.0 mg mL−1) and Ca2+ levels (1.0–10 mM), such as at 2.59 ± 0.50 mM, 4.33 ± 0.41 mM, and 5.07 ± 0.33 mM Ca2+ for 2.0 mg mL−1, 4.0 mg mL−1, and 6.0 mg mL−1 SA, respectively. These results were consistent with FTIR, SAXS, and AFM data. The FTIR spectra showed red shifts and splitting of the carboxylate (–COO−) band around 1600 cm−1, signifying progressive cross-linking into the intermediate stage prior to gel formation. SAXS analysis revealed fiber aggregation at critical Ca2+ concentrations, with a sharp increase in the cylinder radius observed between 2.0–4.0 mM (2.0 mg mL−1 SA) and 4.0–6.0 mM (4.0 and 6.0 mg mL−1 SA). AFM imaging confirmed the three-phase model: early-stage nucleation with fibril formation at low Ca2+, aggregation into dense fibers at intermediate Ca2+ concentrations, and the development of a 3D entangled network in the gelation phase (SA > 6.0 mg mL−1, Ca2+ > 6.0 mM). Overall, CD spectroscopy proved to be a highly sensitive and easy technique for detecting early assembly transitions in the non-gel phase and effectively complemented FTIR, SAXS, and AFM in mapping the SA–Ca2+ assembly process.
Tatsuki Haga, Satoshi Hashimoto, Masaya Yoshida, Yudai Ono, Takeharu Haino, Yasuyuki Maki, Koichi Matsuo, Mohamed I. A. Ibrahim Soft Matter, 2026, 22, 1033-1043.
2025


14. A chiroptical switcher. Helical inversion of twisted porphyrin macrocycle synergistically controlled by combined stimuli of solvent and pressure

Herein, we report the unprecedented discovery of 1 that possesses dynamically tunable chiroptical properties under hydrostatic pressure. Upon hydrostatic pressurization, the g factor inverts from negative to positive. These chiroptical switches are based on pressure-induced conformational change governed by the interplay between macrocyclic flexibility and solvent polarity.
Tomoyuki Hamachi, Tomokazu Kinoshita, Kouta Tanabe, Naoyuki Hisano, Takeharu Haino, Gaku Fukuhara, Chem. Commun., 2025, 61, 18340-18343.(Back Cover)

13. Twisted Host–Guest Systems Exhibit Superamplification of Asymmetry via Coupled Strong Association and Helicity-Driven Cooperativity

Chiral host–guest systems are central to applications in chiral separation, chiral sensing, and asymmetric catalysis. Chiral interactions involving multiple guest molecules exhibit three key features: helicity-driven amplification of asymmetry (i.e., chiral amplification), mutually exclusive binding, and extremely high selectivity. Coupling of amplification of asymmetry with the latter two features can lead to a phenomenon that surpasses conventional amplification of asymmetry, which we propose to term ‘superamplification of asymmetry’. Despite its potential, no established theoretical framework exists. In this work, we developed three models: a general 1:2 twisted host–guest interaction model; the 1:n model assuming independent binding events at multiple sites; and a model assuming mutually exclusive binding. These models were applied to twisted macrocycles and triple-stranded metallohelicates, offering new insights into the amplification of asymmetry mechanisms. Superamplification of asymmetry unlocks powerful new strategies for chiral separation, asymmetric catalysis, and sensing through highly cooperative molecular interactions.
Nozomu Suzuki, Kazuya Miyata, Yudai Ono, Yutaro Yamasaki, Takeharu Haino, J. Am. Chem. Soc., 2025, 147, 43683-43691.

12. Single Crystal Structures and Photophysical Properties of Brucine Complexes with Axially Chiral Biphenyl Derivatives

Biphenyl derivatives that are substituted at the 2,2',4,4',6,6'-positions exhibit axial chirality due to restricted rotation around the central bond, rendering them crucial chiral sources for various functional materials. The present study reports the optical resolution and crystal structure determination of the brucine complexes (R)-1•brucine and (S)-1•brucine, and the spectroscopic characterization of (R)-1•brucine, (S)-1•brucine, (R)-1, and (S)-1. The optical resolution was achieved through the diastereomeric complexation of rac-1with brucine, and the absolute configurations of (R)-1•brucine and (S)-1•brucine were confirmed via single-crystal X-ray diffraction analysis. In the complexes, the brucine molecule was tightly bonded to (R)-1 and (S)-1 by the ionic intermolecular hydrogen-bonding interactions. The additional intermolecular CH–π interaction is exclusive to be present between the brucine CH and the aromatic ring of (R)-1, which most likely facilitates the crystallization of (R)-1•brucine over (S)-1•brucine, leading to the efficient optical resolution. The use of UV-vis absorption, fluorescence, and circular dichroism (CD) spectroscopy resulted in distinct spectral signatures arising from a difference in the structures of the intermolecular hydrogen-bonding interactions in these complexes. Time-dependent density functional theory (TD-DFT) calculations employing chloroform solvation models (CPCM) successfully reproduced experimental absorption spectroscopies, thereby demonstrating a charge-transfer HOMO–LUMO electron transition from the brucine unit to the biphenyl unit.
Yudai Ono, Yuma Nakamura, Takeharu Haino, J. Chin. Chem. Soc., 2025, 72, 1073-1081
11. Controlled Molecular Orders in Layered Multiple Porphyrins

Nature precisely regulates multicomponent assemblies with the assistance of cooperativity. However, establishing such high precision in multicomponent assemblies of artificial supramolecular structures remains challenging. Here, we successfully position multiple distinct guest molecules within two equivalent binding cavities of a zinc-metallated trisporphyrin host by combining two distinct negative cooper-ative interactions, including donor–acceptor π-stacking and metal–ligand coordination. Comprehensive characterization using UV-Vis absorption spectroscopy and diffusion-ordered NMR spectroscopy confirmed the exclusive formation of a ternary supramolecular com-plex. X-ray crystallographic analysis further revealed that the introduction of an additional bridging ligand effectively linked the two ternary complexes to produce an unprecedented septenary supramolecular assembly with alternating guest sequences. In contrast to conventional methods, which require structural differentiation or positive cooperativity, our strategy relies exclusively on negative cooperativity to achieve highly precise molecular ordering. This study presents a novel approach toward constructing sophisticated multicomponent molecular assemblies, emphasizing the significant but underutilized role of negative cooperativity in achieving molecular precision in artificial supramolecular chemistry.
Tomoki Kodama, Naoyuki Hisano, Shin-ichi Tate, Takeharu Haino, J. Am. Chem. Soc., 2025, 147, 31671-31678.
10. Vortex-Flow-Directed Chiral Macroscopic Ordering of Platelet Nanostructures Formed via the Supramolecular Assembly of Platinum Complexes with Bis(phenylisoxazolyl)benzene

To understand the vortex flow-directed circular dichroism (CD) effect observed in homogeneous solutions containing supramolecular structures, the macroscopic order formed by supramolecular structures oriented within a flow must be visualized. In this study, a bis(phenylisoxazolyl)benzene-attached platinum complex was found to self-assemble to form uniform anisotropic platelet nanostructures that are oriented within a flow, thereby generating a chiral macroscopic order that is responsible for CD and linear dichroism (LD) effects only in the vortex flow regime. Cooperative self-assembly of a bis(phenylisoxazolyl)benzene-attached platinum complex via controlled supramolecular polymerization produced anisotropic platelet nanostructures with a narrow polydispersity index. The orientational order parameter of the nanostructures correlated with the flow velocity; thus, the nanostructures were oriented along the flow direction. Furthermore, the vortex flow of the dilute nanostructure solution broke the symmetry of the flow, thereby generating a chiral macroscopic order. As a result, CD and LD effects were observed in the vortex flow regime of the dilute nanostructure solution. These results can be generalized to the formation of chiral macroscopic order in solutions containing anisotropic nanostructures.
Masaya Yoshida, Kyota Yasuda, Takuma Matsumoto, Yudai Ono, Naoyuki Hisano, Mao Kawasaki, Takehiro Hirao, Ye Yuan, Shin-ichi Tate, Martin Vacha, Takeharu Haino, J. Am. Chem. Soc., 2025, 147, 30674-30683.
9. Mechano-responsive color changes of a Pt(ii) complex possessing triethylene glycol towards pressure sensors
Mechanochromic molecules have attracted significant attention owing to their potential in the development of pressure sensors. However, relatively few studies have investigated the detailed mechanisms of the mechano-responsive nature and the quantitative visualization of mechanical forces. Herein, we report a square-planar platinum complex possessing triethylene glycol chains that exhibits mechanocromic behavior in the amorphous phase. Its mechanochromic nature was established using a combination of spectroscopic techniques, powder X-ray diffraction analyses, and computational chemistry techniques. The continuous changes in emission intensity allowed the platinum complex to be used as a mechanical force sensor, where the output signals were readable using a luminescence spectrometer. These findings demonstrate the potential benefits of square-planar platinum complexes and triethylene glycol chains for the creation of mechanochromic material.
Masaya Yoshida, Takehiro Hirao, Shin-ichi Kihara, Takeharu Haino, RSC Adv., 2025, 15, 21401–21407
8. Supramolecular Aggregates of Amide- and Urea-Functionalized Nanographene
Controlling the morphology of supramolecular nanographene(NG) aggregates is challenging. This study confirms that amideand urea-functionalized NG undergo self-assembly to form supramolecular aggregates with a morphology that depends on the incorporated functional group. Amide-functionalized NG forms stacked aggregates, whereas urea-functionalized NG organizes into network polymers. These distinct morphologies suggest that amide groups drive NG stacking, whereas urea groups support NG vertically and horizontally, likely owing to differences in the strengths of single and bifurcated N─H/O hydrogen bonds. Moreover, the functional group incorporated into NG influences the gelation properties of the system. Among the two tested systems, only urea-functionalized NG formed organogels, possibly because urea–urea hydrogen bonds, enable solvent-molecule trapping inside the network polymers formed in these NG systems. Thus, hydrogen bonds can regulate the morphology and function of supramolecular NG aggregates.
Haruka Moriguchi, Ryo Sekiya, Takeharu Haino, ChemistryEurope, 2025, 3, e2500015.
doi.org/10.1002/ceur.202500015
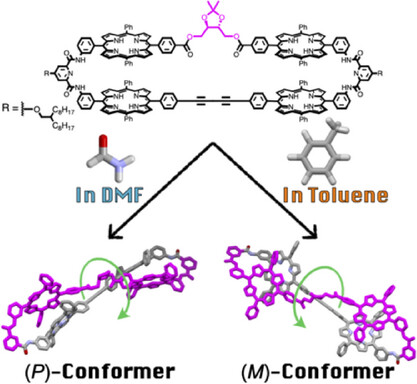
7. Solvent-Directed Handedness in a Chirally Twisted Tetrakisporphyrin Macrocycle
A chirally twisted tetrakisporphyrin macrocycle was synthesized by incorporating a chiral dioxolane into a tetrakisporphyrin macrocycle. The solvent type influenced the preferred handedness of the twisted conformation. Circular dichroism measurements and computational analyses determined the handedness of the conformers in solvents toluene and dimethylformamide.
Kouta Tanabe, Naoyuki Hisano, Takeharu Haino, Asian J. Org. Chem. 2025, 14, e00251.
doi.org/10.1002/ajoc.202500251
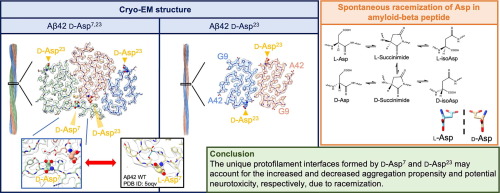
6. Impacts of D-aspartate on the Aggregation Kinetics and Structural Polymorphism of Amyloid β Peptide 1–42
Isomerization of L-Aspartate (L-Asp) into D-aspartate (D-Asp) occurs naturally in proteins at a rate that is much faster than that of other amino acid types. Accumulation of D-Asp is age-dependent, which could alter protein structures and, therefore, functions. Site-specific introduction of D-Asp can accelerate aggregation kinetics of a variety of proteins associated with misfolding diseases. Here, we showed by thioflavin T fluorescence that the isomerization of L-Asp at different positions of amyloid β peptide 1–42 (Aβ42) generates opposing effects on its aggregation kinetics. We further determined the atomic structures of Aβ42 amyloid fibrils harboring a single D-Asp at position 23 and two D-Asp at positions 7 and 23 by cryo-electron microscopy helical reconstruction – cross-validated by cryo-electron tomography and atomic force microscopy – to reveal how D-Asp7 contributes to the formation of a unique triple stranded amyloid fibril structure stabilized by two threads of well-ordered water molecules. These findings provide crucial insights into how the conversion from L- to D-Asp influences the aggregation propensity and amyloid polymorphism of Aβ42.
Li-Ching Hsiao, Chih-Hsuan Lee, Karine Mazmanian, Masaya Yoshida, Genta Ito, Takuya Murata, Naoko Utsunomiya-Tate, Takeharu Haino, Shih-ichi Tate, Shang-Te Danny Hsu, Journal of Molecular Biology, 2025, 437, 169092
doi.org/10.1016/j.jmb.2025.169092
5. Helical Supramolecular Polymers Formed via Head-to-Tail Host-Guest Complexation of Chiral Bisporphyrin Monomers with Trinitrofluorenone
The intermolecular host-guest complexation of head-to-tail monomers consisting of cleft-shaped bisporphyrin and trinitrofluorenone units connected by a chiral binaphthyl linker was employed to construct helically twisted supramolecular polymers. Results from 1H NMR, diffusion-ordered NMR spectroscopy, and viscometry experiments revealed that the supramolecular polymerization of these monomers follows a ring-chain competition mechanism. The introduction of bulky substituents at the linker significantly suppressed the formation of macrocyclic oligomers, whereas smaller alkyl chains facilitated the formation of the cyclic form. The chirally twisted structures of the supramolecular polymers were confirmed using circular dichroism spectroscopy. Atomic force microscopy revealed that the (R)- and (S)-configurations of the binaphthyl linkers induced right- and left-handed helical structures, respectively, in the supramolecular polymer chains. The absence of cooperativity in the supramolecular copolymerization of (R)- and (S)-1a resulted in the formation of stereo-random supramolecular copolymers.
Naoyuki Hisano, Tomoki Kodama, Soichiro Koya, Takeharu Haino, Chem. Eur. J., 2025, 31, e202404210.(Very Important Paper, Front Cover)
4. Chirality Generation on Carbon Nanosheets by Chemical Modification
Chirality is an intriguing property of molecules, and an exciting area of study involves the generation of chirality in nanographene (NGs), also known as graphene quantum dots. Unlike those synthesized through stepwise carbon-carbon bond formation by organic reactions (bottom-up method), NGs are obtained by cutting parent carbons (top-down method) pose challenges in precisely regulating their three-dimensional structures by post-synthesis. This includes the incorporation of non-hexagonal rings and helicene-like structures in carbon frameworks. Currently, edge functionalization is the only method for generating chirality in NGs produced by the top-down method. While various chiral NGs have been synthesized through organic methods, examples of chemical modification remain rare due to limited structural information and the substantial size of NGs. However, these problems can be mitigated by disclosing the structures of NGs, particularly their edge structures. This minireview focuses on recently published papers that address the structural characterization of NGs and their chirality generation by edge modification. Comparing these NGs with those synthesized by organic synthesis will help to develop reasonable strategies for creating sophisticated chiral NGs. We hope this mini-review contributes to the advancement of NG-organic hybrid materials.
Ryo Sekiya, Saki Arimura, Haruka Moriguchi, Takeharu Haino, Nanoscale, 2025, 17, 774-787.
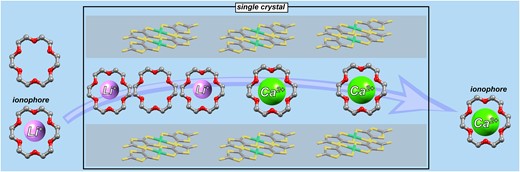
3. Single-crystal-to-single-crystal transformation based on ionophore-like transport
We have already reported that Li2([18]crown-6)3[Ni(dmit)2]2(H2O)4 crystals, which have an ion channel structure, can be reversibly converted to Ca([18]crown-6)[Ni(dmit)2]2(H2O)3 crystals by ion exchange while maintaining their single-crystal state. In this process, inorganic ions as well as crown ethers traverse in and out of the crystal. In this study, [18]crown-6-d4, a partially deuterated [18]crown-6, was used to investigate the behavior of inorganic ions and crown ethers during ion exchange and found that Li+-[18]crown-6 supramolecules could act as ionophores in the crystal.
Mizuki Ito, Jun Manabe, Katsuya Inoue, Takehiro Hirao, Takeharu Haino, Tomoyuki Akutagawa, Kiyonori Takahashi, Takayoshi Nakamura, Sadafumi Nishihara, Chem. Lett., 2025, 54, upae252.
doi.org/10.1093/chemle/upae252
2. Controlled Helical Organization in Supramolecular Polymers of Pseudo-Macrocyclic Tetrakisporphyrins

Tetrakisporphyrin monomers with amino acid side chains at each end form intramolecular antiparallel hydrogen-bonds to adopt chirally twisted pseudo-macrocyclic structures that result in right-handed and left-handed (P)- and (M)-conformations. The pseudo-macrocyclic tetrakisporphyrin monomers self-assembled to form supramolecular helical pseudo-polycatenane polymers via head-to-head complementary dimerization of the bisporphyrin cleft units in an isodesmic manner. The formation of one-handed supramolecular helical pseudo-polycatenane polymers was confirmed by circular dichroism spectroscopy. The methyl and iso-propyl groups at the stereogenic center greatly enhanced the induced circular dichroism (CD) in the Soret bands of the supramolecular helical pseudo-polycatenane polymers. The induced CDs were reduced upon the introduction of large iso-butyl and tert-butyl groups. Atomic force microscopy revealed well-grown and long supramolecular helical pseudo-polycatenane polymer chains with chain lengths in the range of 361 to 13.6 nm. The right-handed helical chains were established by the self-assembly of the right-handed (P)-conformation of the pseudo-macrocyclic monomer.
Naoka Fujii, Naoyuki Hisano, Takehiro Hirao, Shin-ichi Kihara, Kouta Tanabe, Masaya Yoshida, Shin-ichi Tate, Takeharu Haino, Angew. Chem. Int. Ed. 2025, 64, e202416770(Hot Paper, Inside Back Cover)
1. Temperature-Dependent Left- and Right-Twisted Conformational Changes in 1:1 Host-Guest Systems: Theoretical Modeling and Chiroptical Simulations
An efficient strategy for high-performance chiral materials is to design and synthesize host molecules with left- and right- (M- and P-) twisted conformations and to control their twisted conformations. For this, a quantitative analysis is required to describe the chiroptical inversion, chiral transfer, and chiral recognition in the host-guest systems, which is generally performed using circular dichroism (CD) and/or proton nuclear magnetic resonance (1H-NMR) spectroscopies. However, the mass-balance model that considers the M- and P-twisted conformations has not yet been established. In this study, we derived the novel equations based on the mass-balance model for the 1:1 host-guest systems. Then, we further applied them to analyze the 1:1 host-guest systems for the achiral calixarene-based capsule molecule, achiral dimeric zinc porphyrin tweezer molecule, and chiral pillar[5]arene with the chiral and/or achiral guest molecules by using the data obtained from the CD titration, variable temperature CD (VT-CD), and 1H-NMR experiments. The thermodynamic parameters (ΔH and ΔS), equilibrium constants (K), and molar CD (Δε) in the 1:1 host-guest systems could be successfully determined by the theoretical analyses using the derived equations.
Nozomu Suzuki, Daisuke Taura, Yusuke Furuta, Yudai Ono, Senri Miyagi, Ryota Kameda, Takeharu Haino, Angew. Chem. Int. Ed. 2025, 64, e202413340.